
BatchTrak™: Regulatory Alignment with ISPE and GAMP 5
Document Control
Document Title
BatchTrak™ System Impact Assessment Whitepaper
Document Number
ENG-102043
Version
1.1
Author
GMPKit, LLC.
Approval Authority
GMPKit Quality & Compliance
Original Publication Date
October 2025
Document Purpose
This whitepaper summarizes the System Impact Assessment (SIA) performed for the BatchTrak™ platform and explains how the system aligns with recognized regulatory guidance for computerized systems used in regulated pharmaceutical manufacturing environments.
Executive Summary
Batch-based pharmaceutical and biologics manufacturing environments rely on a complex ecosystem of computerized systems to manage production, quality, and compliance activities. Before implementing new software within regulated environments, organizations must evaluate whether the system impacts product quality, patient safety, or regulatory compliance.
To support responsible deployment in regulated manufacturing environments, GMPKit conducted a formal System Impact Assessment (SIA) for the BatchTrak™ platform using internationally recognized regulatory guidance including:
- ISPE Baseline Guide Volume 5 — Commissioning & Qualification
- ISPE GAMP 5 — A Risk-Based Approach to Compliant Computerized Systems
- FDA 21 CFR Part 11 — Electronic Records and Electronic Signatures
- EU GMP Annex 11 — Computerized Systems
- ISO 27001 — Information Security Management
The purpose of this assessment was to determine:
- Whether BatchTrak™ impacts product quality or patient safety
- The appropriate system classification under ISPE guidance
- The appropriate level of commissioning or validation activities
Based on the risk evaluation framework defined by ISPE, BatchTrak™ is classified as a procedural support platform that does not directly impact product quality, patient safety, or GMP compliance decisions.
BatchTrak™ operates within the operational coordination layer of the manufacturing technology stack, integrating with — but not replacing — validated GxP systems such as MES, LIMS, and QMS platforms.
This whitepaper summarizes the evaluation approach and explains how BatchTrak™ aligns with established regulatory guidance for computerized systems used in regulated manufacturing environments.
The conclusions presented in this whitepaper are derived from the formal System Impact Assessment performed for BatchTrak™ and documented under GMPKit document ENG-102043. The assessment evaluated system boundaries, operational functionality, and potential regulatory impact using the risk-based classification criteria defined within ISPE guidance for computerized systems.
Why System Impact Assessments Matter in Regulated Manufacturing
Regulated pharmaceutical manufacturing environments operate under strict regulatory frameworks designed to ensure product quality, patient safety, and data integrity.
When new computerized systems are introduced into these environments, organizations must determine the appropriate level of regulatory oversight and validation required for that system.
Industry guidance from ISPE recommends performing a System Impact Assessment (SIA) to determine whether the system:
- Directly impacts product quality or patient safety
- Indirectly supports regulated processes
- Has no impact on regulated GMP activities
This classification determines whether the system requires full GxP validation, risk-based commissioning, or internal assurance controls.
By performing this analysis early in the lifecycle, organizations can ensure:
- Appropriate validation scope
- Efficient use of compliance resources
- Alignment with GAMP 5 risk-based principles
GMPKit applies this methodology during the development of its platforms to ensure they can be responsibly deployed within regulated manufacturing environments.
Evaluation Methodology
The BatchTrak™ System Impact Assessment followed a structured evaluation process aligned with ISPE and GAMP guidance.
The assessment included the following steps:
- Definition of system boundaries and intended use
- Identification of interactions with regulated systems
- Evaluation against the ISPE eight-question system classification framework
- Risk evaluation for product quality, patient safety, and data integrity
- Determination of the appropriate GAMP 5 software category
- Definition of the commissioning and qualification strategy
This methodology ensures the system classification reflects both the technical architecture and the operational role of the platform within the manufacturing ecosystem.
This evaluation approach aligns with the risk-based lifecycle principles defined in GAMP® 5 for computerized systems used in regulated manufacturing environments.
BatchTrak™ Platform Overview
BatchTrak™ is a Manufacturing Operations Management (MOM) platform designed to improve operational coordination and visibility across batch-based manufacturing processes.
The platform provides:
- Workflow coordination
- Operational dashboards and reporting
- Task tracking and escalation
- Operational visibility across departments
- Integration with enterprise systems
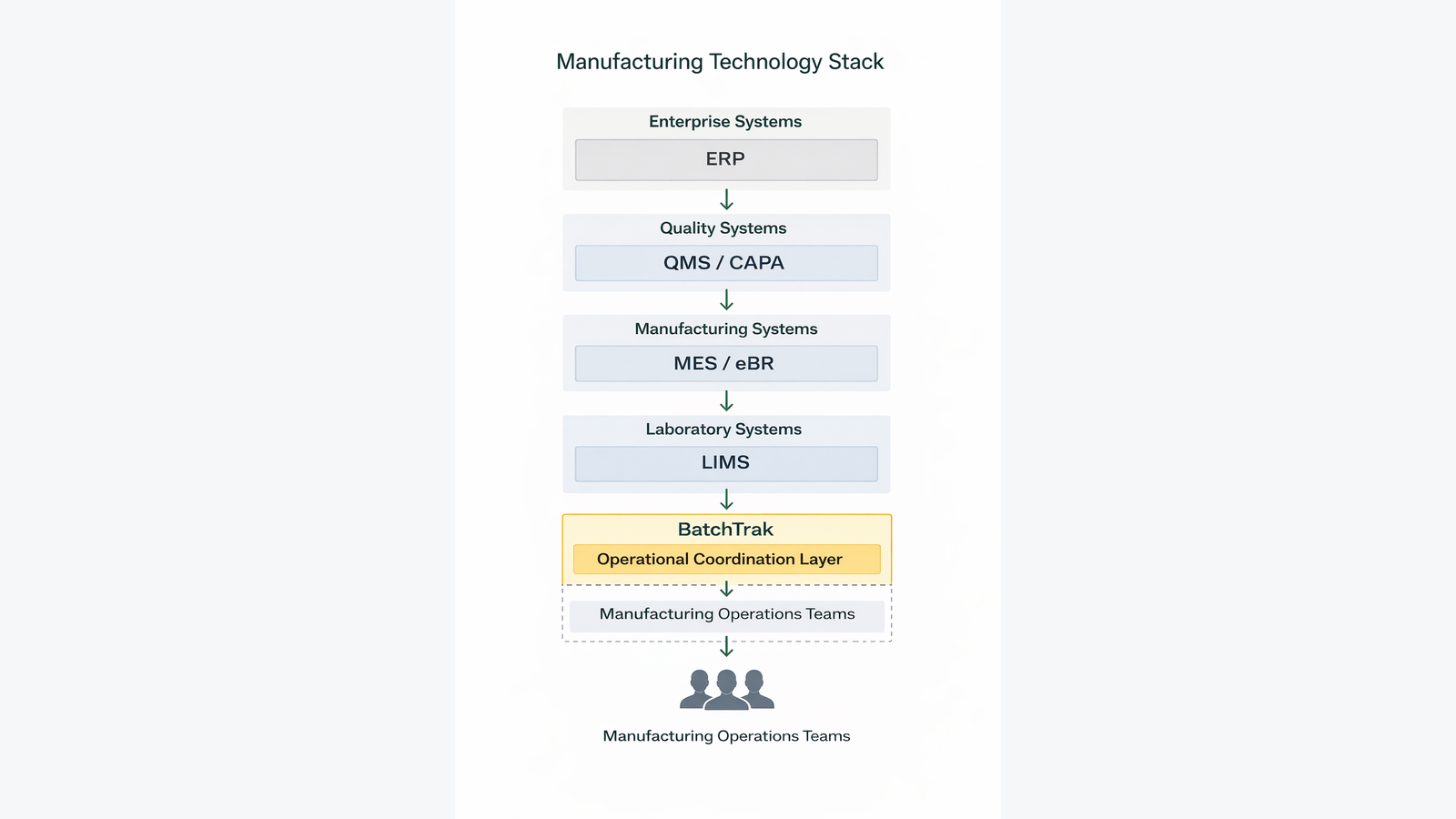
BatchTrak™ operates in the information coordination layer of the manufacturing technology stack and integrates with enterprise systems through secure APIs.
Importantly, the platform does not replace validated GxP systems responsible for managing product quality data or executing regulated processes.
System Boundary Definition
Defining system boundaries is critical when evaluating regulatory impact.
The following boundaries were established during the assessment:
In-Scope Components (Managed by GMPKit)
BatchTrak™ includes:
- Web application user interface
- Workflow orchestration engine
- Operational dashboards and reporting
- User authentication and access control
- API gateway for system integrations
- Notification and audit trail services
These components support operational coordination and information visibility.
Out-of-Scope Systems
BatchTrak™ integrates with — but does not replace — validated enterprise systems, including:
- Manufacturing Execution Systems (MES)
- Electronic Batch Record systems (eBR)
- Laboratory Information Management Systems (LIMS)
- Quality Management Systems (QMS)
- Enterprise Resource Planning (ERP)
- Process control systems (DCS, SCADA, PLC)
Product quality decisions and GMP records remain within these validated systems.
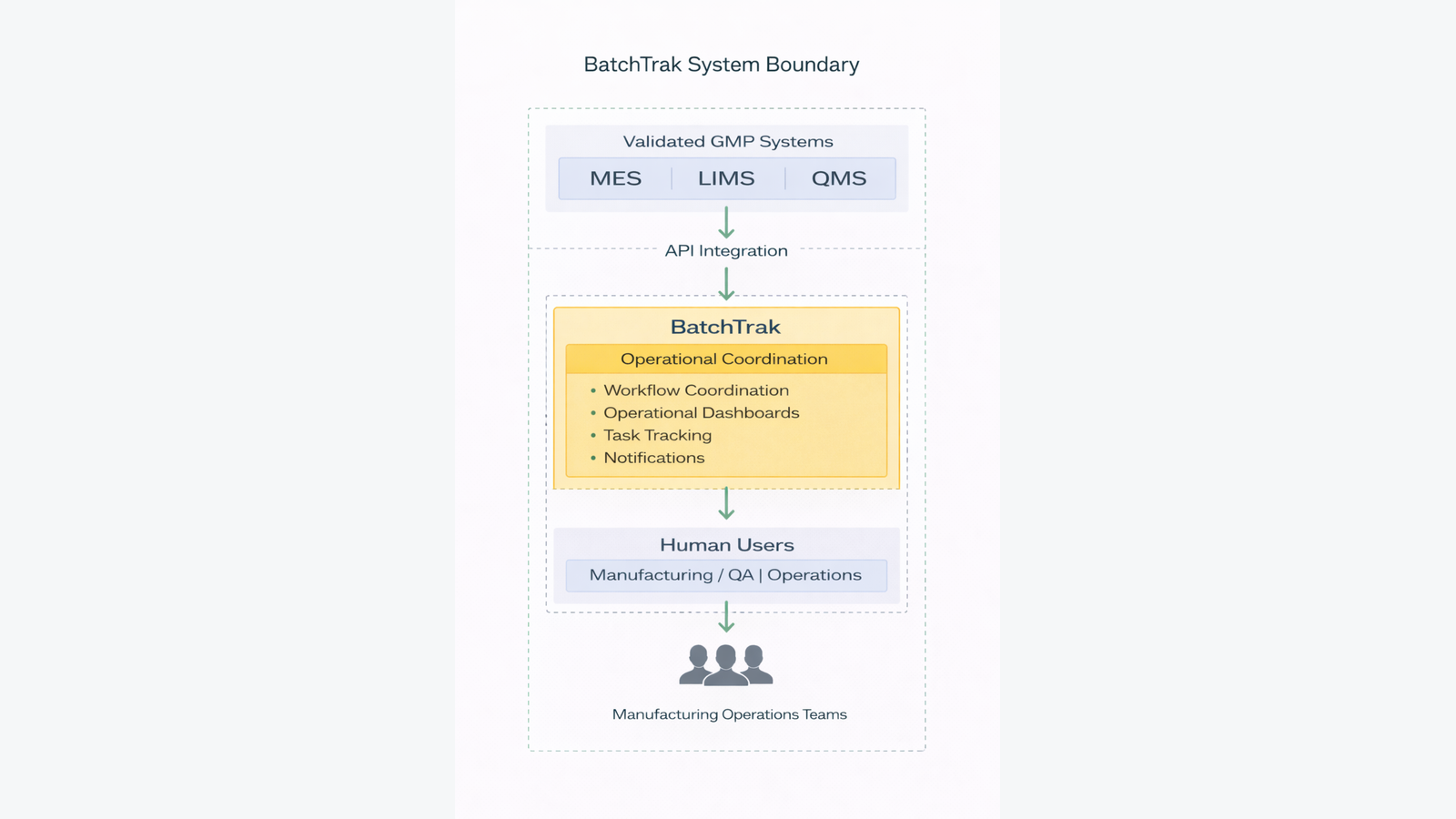
BatchTrak™ operates as an operational coordination platform that improves workflow visibility, task management, and operational communication across manufacturing teams while preserving GMP system authority.
System Classification Outcome
The system was evaluated using the ISPE system classification criteria defined within the ISPE Baseline Guide for Commissioning and Qualification:
- Does not control manufacturing processes
- Does not interact directly with the product or process stream
- Does not perform automated product release decisions
- Does not replace validated quality systems
- Does not generate authoritative GMP records
Instead, BatchTrak™ provides operational coordination and visibility across manufacturing activities.
As a result, the platform is classified as a:
Procedural Support System (Indirect Impact)
This classification means the system supports operational processes but does not directly impact product quality or patient safety.
GAMP 5 Software Category
Under the GAMP 5 framework, BatchTrak™ is categorized as a:
Category 4 – Configured Product
Category 4 systems are:
- Commercial off-the-shelf software platforms
- Configurable to customer workflows
- Managed through supplier lifecycle documentation
- Not modified through custom source-code changes by end users
This classification aligns with the BatchTrak™ architecture, which allows organizations to configure workflows and operational processes without modifying the underlying platform codebase.
Risk Management Approach
A risk-based evaluation was performed to assess potential impacts across key operational and compliance domains.
The assessment concluded:
Product Quality — Low Risk
BatchTrak does not control manufacturing processes, interact with product streams, or make automated product release decisions.
Patient Safety — Low Risk
The platform does not interact with the product or production environment and therefore cannot directly impact patient safety.
Data Integrity — Medium Risk
Operational data and workflow coordination require appropriate controls such as role-based access, audit trails, and secure tenant isolation.
Operational Risk — Medium Risk
System availability and performance are important for operational coordination but do not directly affect regulated manufacturing execution.
Risk mitigation controls include:
- Role-based access control (RBAC)
- Comprehensive audit trail capabilities
- Secure SaaS architecture
- Tenant data isolation
- Disaster recovery and high-availability infrastructure
These controls support secure and reliable operation within enterprise manufacturing environments.
Commissioning and Qualification Strategy
Based on the system classification and risk evaluation, BatchTrak™ follows a risk-based commissioning approach aligned with GAMP 5 principles.
The recommended commissioning strategy includes:
Installation Qualification (IQ)
Verification that the system architecture and infrastructure are deployed according to design specifications, including:
- Platform architecture
- Security configuration
- Tenant isolation controls
- Network configuration
Operational Qualification (OQ)
Testing of configured system functionality including:
- Workflow orchestration
- Status transitions
- Access controls
- Audit trail functionality
- Error handling
Performance Qualification (PQ)
User acceptance testing in a production-like environment to confirm that the platform performs consistently under expected operational conditions.
Implications for Regulated Manufacturers
The BatchTrak™ architecture is intentionally designed to operate alongside validated manufacturing and quality systems rather than replacing them.
This design approach allows organizations to improve operational coordination and visibility without introducing unnecessary regulatory complexity.
By maintaining clear system boundaries and preserving GMP decision-making within validated systems, BatchTrak™ can be deployed using a risk-based commissioning strategy aligned with GAMP 5 guidance.
Regulatory References
The BatchTrak™ System Impact Assessment was conducted using internationally recognized regulatory and industry guidance for computerized systems used in regulated manufacturing environments.
Primary references include:
ISPE Baseline Guide Volume 5 — Commissioning & Qualification
International Society for Pharmaceutical Engineering (ISPE)
ISPE GAMP® 5 — A Risk-Based Approach to Compliant Computerized Systems
Second Edition
FDA 21 CFR Part 11 — Electronic Records; Electronic Signatures
EU GMP Annex 11 — Computerized Systems
ISO/IEC 27001 — Information Security Management Systems
These standards provide a risk-based framework for determining the appropriate level of validation and control required for computerized systems used in regulated pharmaceutical manufacturing environments.
Conclusion
GMPKit applies structured regulatory evaluation methods during the development of its platforms to ensure they can be responsibly deployed within regulated manufacturing environments.
The System Impact Assessment for BatchTrak™ determined that the platform functions as a procedural support system that does not directly impact product quality, patient safety, or GMP compliance decisions.
As a result, BatchTrak™ aligns with GAMP 5 principles for configurable software platforms and can be implemented using a risk-based commissioning approach appropriate for its operational role.
This approach enables regulated manufacturers to gain improved operational visibility and coordination while maintaining compliance with established industry guidance for computerized systems.
About GMPKit
GMPKit is a specialist consultancy and software provider focused on improving execution stability and reducing the Cost of Poor Quality (COPQ) in batch-based pharmaceutical and biologics manufacturing environments.
The GMPKit platform ecosystem includes:
BatchTrak™ – A Manufacturing Operations Management platform designed to improve operational coordination and visibility across batch-based manufacturing processes.
GMPWit™ – An AI-assisted knowledge platform designed to support GMP decision-making, deviation investigation guidance, and operational learning within regulated environments.
GMPKit combines operational consulting, digital platforms, and leadership training to help organizations restore execution stability and recover hidden operational losses across manufacturing operations.
Learn more at www.gmpkit.com
