
How to Calculate Your Facility's True COPQ (Free Calculator)
Understanding the Cost of Poor Quality (COPQ) in Biopharma
In quality engineering, the Cost of Poor Quality (COPQ) refers to all the financial losses an organization incurs when products, processes, or services fall short of quality standards.
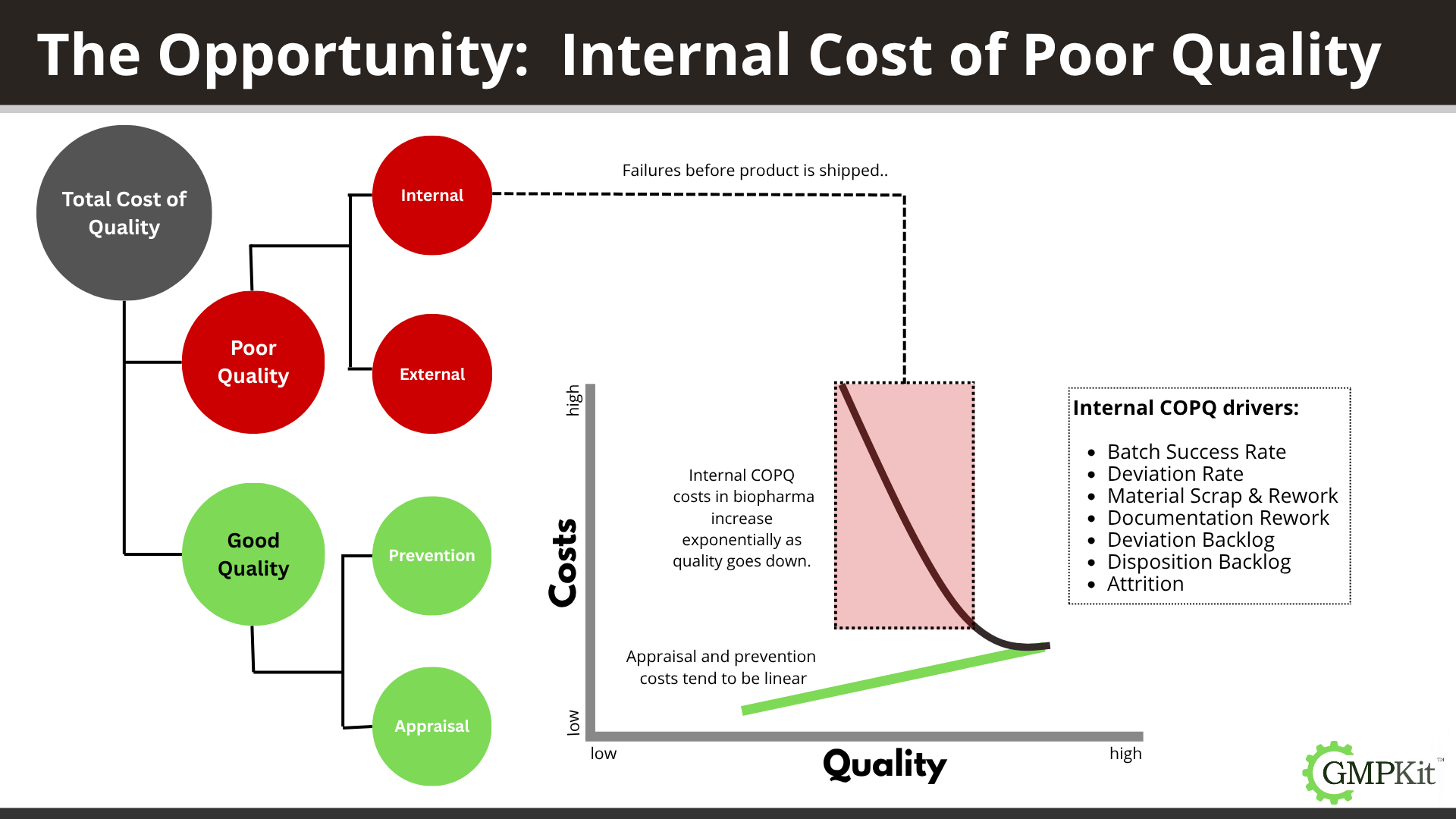
Internal failure consequences can include product rejections and batch failure, deviation investigations, raw material loss, manufacturing rework, documentation errors and correction, and even attrition. These costs add up quickly.
Despite these huge risks, many biopharma organizations underestimate just how much poor quality is costing them—sometimes up to 20% of revenues—hidden in plain sight.
Where Do Quality Costs Show Up in Biopharma?
Let’s break it down. Your overall “cost of quality” can be thought of in terms of investments and failures:
- Prevention is your up-front investment: staff training, SOPs, method validation, equipment calibration, CAPAs, and proactive process risk management. It's all the work you do to make sure things go right the first time.
- Appraisal costs are what you spend testing and verifying your processes—the sampling, QC checks, release testing, and audits that ensure compliance and performance.
- Internal Failure comes when defects are detected before product leaves your facility: think failed batches, deviations, OOS investigations, or discarded intermediates.
- External Failure happens when a problem escapes into the market—field complaints, recalls, regulatory warning letters, and customer remediation. These are the most damaging and expensive failures in regulated biopharma.
Why Does COPQ Skyrocket in Biopharma?
Quality failures in biopharma are more costly and complex than in most industries due to:
- Strict global regulatory oversight
- High-value inventories and extended production cycles
- Complex biological processes
- Consequential impact on patient health and safety
- The logistics and reputational fallout of recalls and supply interruptions
Small investments in prevention and testing (like extra method robustness studies, tech transfer controls, real time batch tracking, or digital QMS upgrades) often cost a fraction of what a single recall, public incident, or even repeated batch rejection would drain from your business. The further a defect gets—especially out to the patient—the more exponentially the costs explode, including regulatory interventions, remediation, and long-term brand damage.
Making the Case for Calculating COPQ
When you know your true COPQ numbers, you can:
- Expose the hidden losses undermining your margins and competitiveness
- Justify investment in quality improvement to stakeholders and leadership
- Pinpoint high-risk processes or products in your pipeline
- Measure and sustain improvements in real time
In a field where “right first time” and full compliance are non-negotiable, understanding and reducing your COPQ translates directly to better patient outcomes, fewer regulatory headaches, and a healthier bottom line.
Free, Simple COPQ Calculator for Biopharma
Ready to see your facility’s real internal COPQ? Try GMPKits Cost of Poor Quality Calculator—a free, user-friendly tool designed for biopharma facilities.
Input your actual batch success rate, deviation rates, scrap and rework to get a clear picture of where your quality dollars are going, how you stack up against industry norms, and how much you could save by investing in prevention and appraisal.
Take the guesswork out of quality investments. Try the free calculator and start unlocking potential savings—and peace of mind—for your biopharma operation today.
Tags
Related Posts

How To Write the Perfect Problem Statement
If you work in Biopharmaceutical Manufacturing or Quality, one of the best things you can do to drive reliability in GMP Manufacturing is learn how to craft a strong problem statement. In this video, we will show you how to do that. Creating a strong problem statement is pivotal in problem-solving. A well-defined problem statement sets the stage for effective GMP deviation investigation and problem solving.

What is A GMP Deviation Investigation?
In GMP pharmaceutical production, including biologics and cell therapy manufacturing, a GMP Deviation Investigation is the structured process used to identify, document, analyze, and resolve any unexpected events or conditions that do not comply with approved procedures, specifications, or regulatory requirements.

BatchTrak - Is it an Obeya Room?
What is an Obeya Room? An Obeya room (from the Japanese word "Obeya" 大部屋, meaning "big room") is a centralized, physical or digital space used for collaborative project management and visual coordination, especially in Lean and Agile environments.
